gamma.-Aminobutyric acid esters. I. Synthesis, brain uptake, and pharmacological studies of aliphatic and steroid esters of .gamma.-aminobutyric acid | Journal of Medicinal Chemistry

Exploitation of Antibiotic Resistance as a Novel Drug Target: Development of a β-Lactamase-Activated Antibacterial Prodrug | Journal of Medicinal Chemistry

Exploitation of Antibiotic Resistance as a Novel Drug Target: Development of a β-Lactamase-Activated Antibacterial Prodrug | Journal of Medicinal Chemistry

Bisphosphonate prodrugs: synthesis and in vitro evaluation of novel partial amides of clodronic acid - ScienceDirect
Application of Intestinal Epithelial Cells Differentiated from Human Induced Pluripotent Stem Cells for Studies of Prodrug Hydro
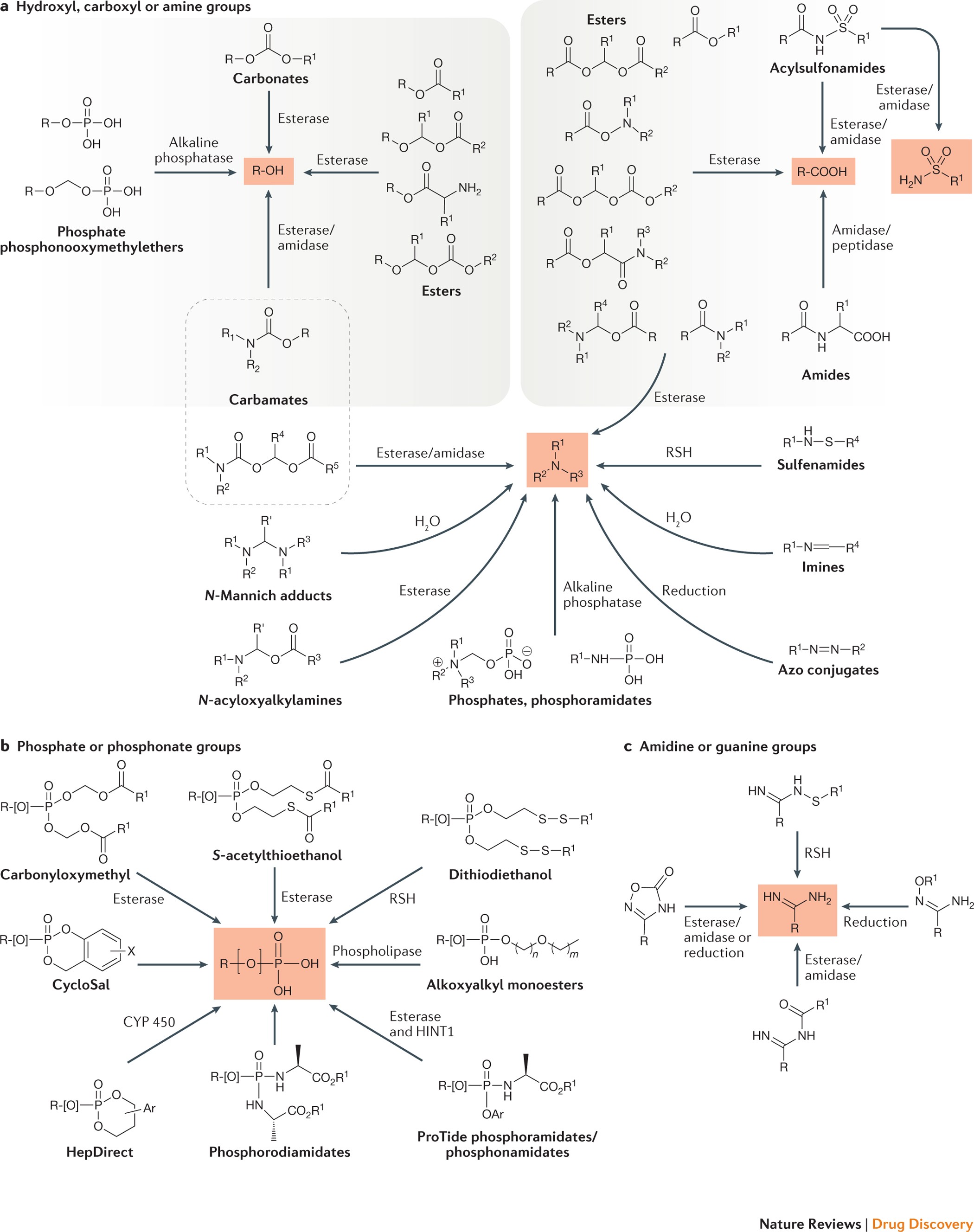
The expanding role of prodrugs in contemporary drug design and development | Nature Reviews Drug Discovery

Self‐Synthesizing Nanorods from Dynamic Combinatorial Libraries against Drug Resistant Cancer - Cao - 2021 - Angewandte Chemie International Edition - Wiley Online Library

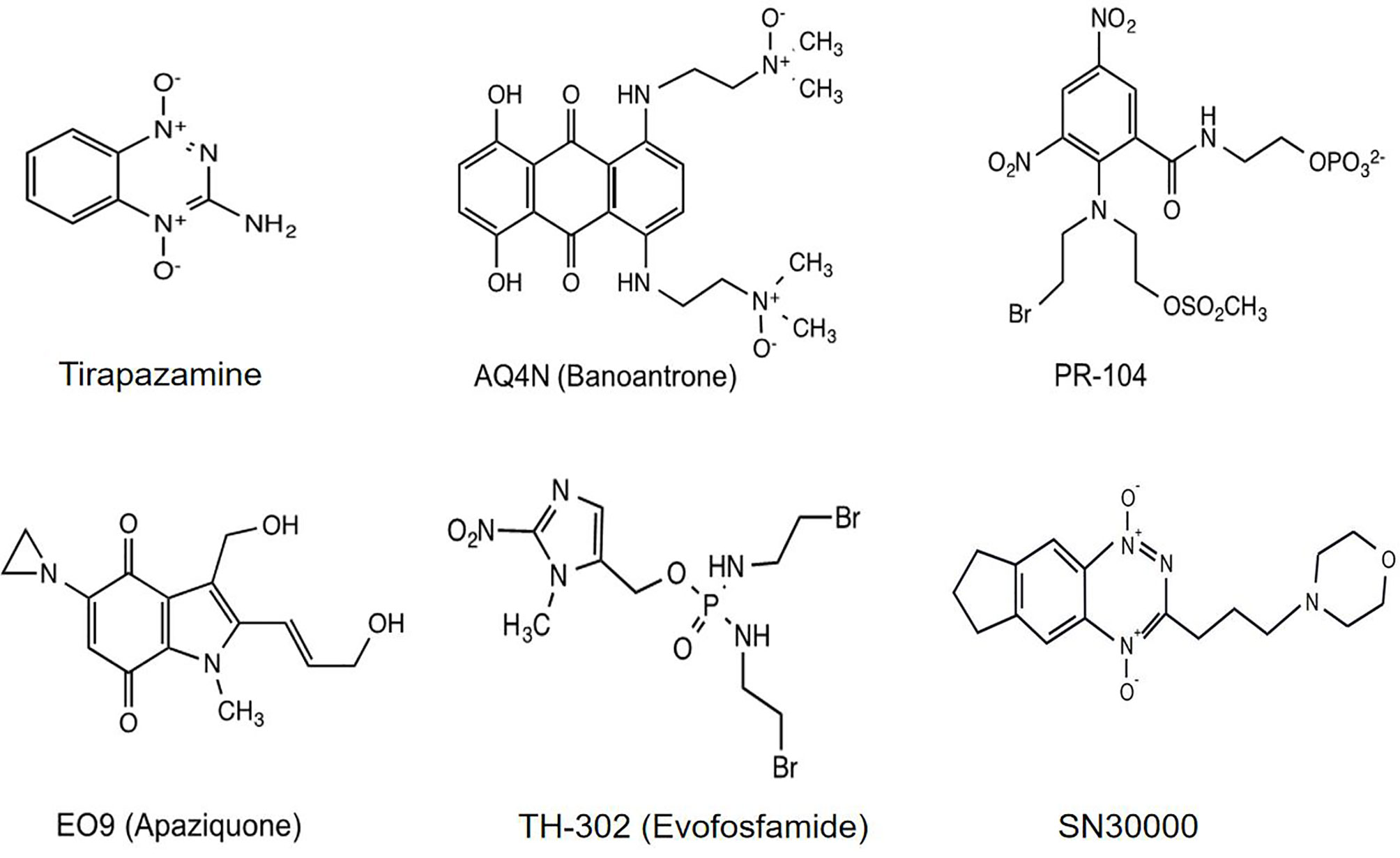
Self‐Synthesizing Nanorods from Dynamic Combinatorial Libraries against Drug Resistant Cancer - Cao - 2021 - Angewandte Chemie International Edition - Wiley Online Library